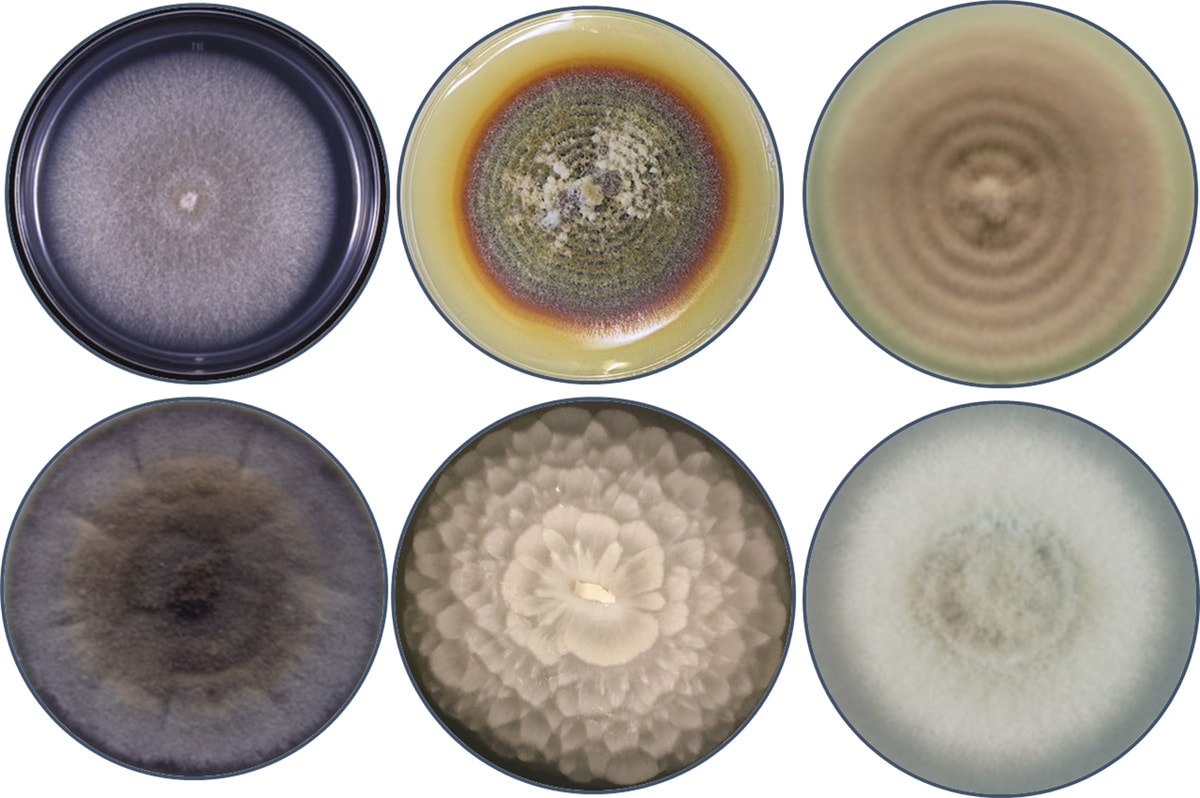
MicroBIOME OF SCN SUPPRESSIVE SOILS AND NeMATOPHAGOUS FUNGI FOR BIOCONTROL OF THE SOYBEAN CYST NEMATODE (SCN)
.With funding from the USDA-NIFA, the Minnesota Soybean Research and Production Council, and the NY Corn and Soybean Growers Association, and in collaboration with nematologists Senyu Chen (University of Minnesota) and Xiaohong Wang (USDA-ARS, Ithaca, NY)we have isolated fungal cultures from soil, roots, and various stages of the soybean cyst nematode (Heterodera glycines; SCN) life-cycle from a long-term corn-soybean rotation experiment at the Southern Research and Outreach Center in Waseca, Minnesota and farms in upstate NY as part of the SCN survey. Research questions include 1) how do fungal and bacterial communities in soil, roots, and SNC cysts change over crop rotation and long-term monoculture in agroecosystems, 2) what key taxa may contribute to the ability to some soils to develop the ability to suppress SCN populations, 3) how do interactions between key community members or 4) enzymatic functions (e.i. CAZymes involved in chitin degradation) or secondary metabolism of microbial players contribute to SCN suppressive soils. In addition to interrogating the SCN and suppressive soil microbiomes for their role(s) in controlling SCN, we are screening individual fungal isolates for the ability to either parasitize or produce metabolites toxic to the SCN and conducting greenhouse screenings to identify candidate strains for biocontrol agents. We aim to identify fundamental mechanisms of parasitism or modes of action against SCN in in-vitro assays and perform bioactivity guided fractionation of filtrates for chemical analyses of highly antagonistic isolates to identify active compounds. In collaboration with other USDA researchers, we are also screening these fungi against other species of cyst (Potato cyst and Golden cyst (Globodera spp.), as well as other agriculturally relevant nematodes (Meloidogyne, Pratylenchus). As part of the Forever Green project at University of Minnesota, we also investigated the endophytic potential of a nematophagous fungus (Pochonia chlamydosporia) to control the SCN in an emerging cover crop species, Pennycress (Thlaspi arvense L.), and its close relative Arabidopsis. To understand the role of endophytic fungi in protecting plants from disease, we are sequencing the genomes of several nematophagous fungi through the Community Sequencing Project (CSP): "Defensive Mutualism of Fungal Root Endophytes of Soybean") at the Joint Genome Institute (JGI) .
Role of Microbes IN THE INVASIVE BIOLOGY OF WOOD BORING BEETLES
The emerald ash borer (EAB;Agrilus plannipennis) is an invasive beetle from Asia that has killed hundreds of millions of North American ash trees over the last two decades. As the developing beetle larvae feed on the tissues of the inner bark, the galleries they excavate effectively sever the connection between the canopy and the roots, rapidly killing the tree. Microbial symbionts of other wood-feeding beetles such as bark- and ambrosia beetles are known to play important roles in metabolizing lignin and detoxifying plant compounds to increase the nutritional value of wood or decrease its toxicity to the insect larvae. Very little is known about microbes associated with the EAB and other destructive congeneric phloem-boring beetles. With funding from NSF-DOB and in collaboration with researchers in China (Dr. Jianghua Sun, Chinese Academy of Sciences) and other researchers in the U.S. (Bob Blanchette, University of Minnesota; Kayla Perry, Ohio State University, Kamal Ghandi, University of Georgia), we are conducting parallel studies of the microbiota associated with EAB and with both native and non-native ash hosts in the native (China) and invasive (U.S.) ranges of this beetle to understand 1) how the microbiome may vary by ash host, 2) the interactions between microbiome and plant phytochemistry, 3) how the microbiome of the beetle may have changed during invasion, 4) what functions EAB associated microbes may confer to their beetle hosts (or holobiont of insect plus associated microbes), and 5) whether entomopathogenic fungi associated with the beetle may be deployed for biological control. Microbial taxa contributing to EAB’s ability to overcome defenses of bark tissues may also have applications in second-generation biofuel production. Bark, branches, and other forestry by-products constitute an abundant and low-cost lignocellulosic feedstock for biofuel production, but result in low fermentable sugar yields due in part to cellulase inhibition by high levels of water-soluble phenolic compounds. Presumably, EAB and its resident microbiome must possess a means to mitigate cellulase inhibition, either by inactivating bark phenolic compounds or expressing cellulases less sensitive to inhibition.
COMPARATIVE GENOMES OF INSECT VIRULENCE AND ENDOPHYTISM IN THE GENUS BEAUVERIA
Beauveria species are known as insect pathogens, many of which have potential for biocontrol of insect agricultural or forest pests. More recently, however, several species of Beauveria have been isolated as endophytes from plants. Using a combination of comparative genomics, molecular genetics, and functional bioassays, we are investigating the role of specific gene families (i.e. secondary metabolites, carbohydrate active enzymes, proteases, and small secreted proteins) in shaping host- specialization on an insect or plant host and their potential role in defending plants against pathogens as fungal "bodyguards". We are interested in understanding what genomic features allow for a flexible fungal lifestyle that encompasses multiple hosts from different kingdoms of life and the potential for use of these entomopathogens as plant-protective endophytes. Our research is using the study system of the invasive lepidopteran Spodoptera frugiperda on tomato, soybean, and corn plant hosts.
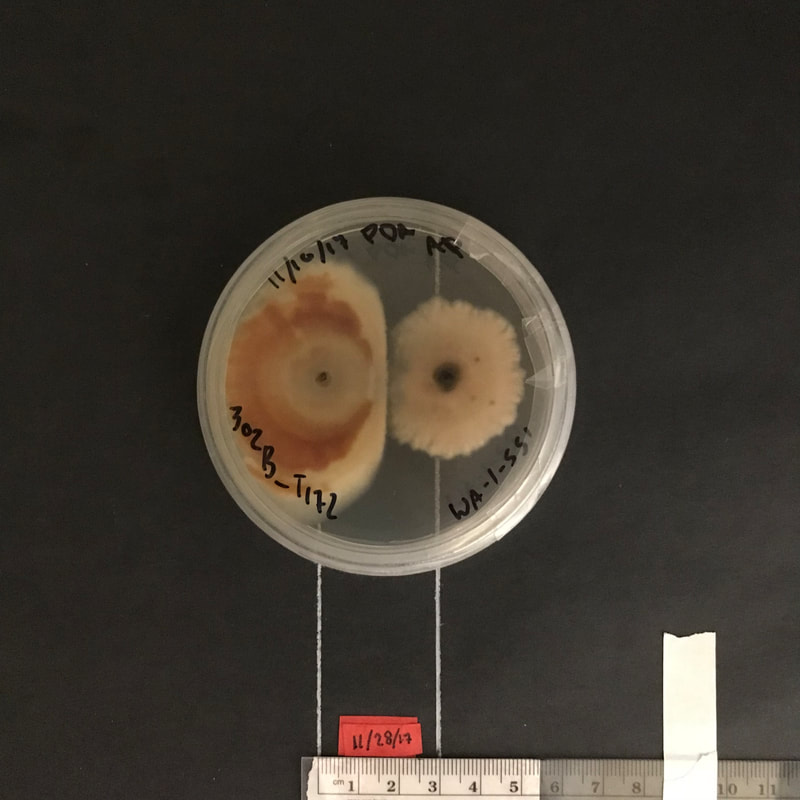
MECHANISMS OF AN INVASIVE FUNGAL PATHOGEN FusariUM VIRGULIFORME
Fusarium virguliforme is the causal agent of the soybean disease sudden death syndrome (SDS), a significant disease of soybean that can also cause root disease on other crops such as dry edible beans, clover, and alfalfa. SDS was first reported in Arkansas in 1971, and by 1993 had spread to Illinois, Indiana, and Iowa, and was detected in Minnesota in 2002. In collaboration with Dean Malvick (University of Minnesota), we are characterizing various traits that may influence its ability as an invasive pathogen to spread, survive, and become a significant pathogen in Minnesota and other areas of the Midwest. These include the ability to compete or antagonize other soil fungi through antibiosis, carbon-use profiles and niche-width, and temperature tolerance. Severity of SDS is often found correlated with the presence of the soybean cyst nematode (SCN), and this project also investigates the role of cysts of the soybean cyst nematode in survival or spread of the fungus.